From January 2020 to October 2020, a prospective methodological study was conducted at the Pisa University Hospital which published on BMC Microbiology. Goldstream® Fungus (1-3)-β-D-Glucan Test was used for detecting the BDG level from BAL samples. The result was quantified by a Fully Automatic Kinetic Tube Reader IGL-200 from Era Biology. The research shows that BDG should be taken into consideration for its high negative predictive value. And it resulted useful in ruling out the diagnosis of PCP for all the negative control patients.

Background:
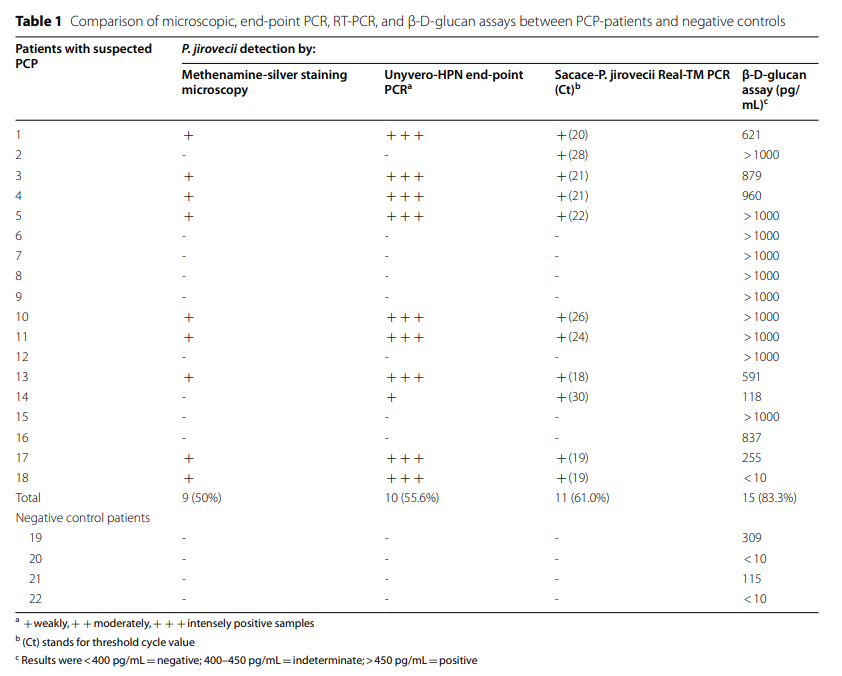
The current diagnostic gold standard for Pneumocystis jirovecii is represented by microscopic visualization of the fungus from clinical respiratory samples, as bronchoalveolar-lavage fluid, defining “proven” P. jirovecii pneumonia, whereas qPCR allows defining “probable” diagnosis, as it is unable to discriminate infection from colonization. However, molecular methods, such as end-point PCR and qPCR, are faster, easier to perform and interpret, thus allowing the laboratory to give back the clinician useful microbiological data in a shorter time. The present study aims at comparing microscopy with molecular assays and beta-D-glucan diagnostic performance on bronchoalveolar-lavage fluids from patients with suspected Pneumocystis jirovecii pneumonia. Bronchoalveolar-lavage fluid from eighteen high-risk and four negative control subjects underwent Grocott-Gomori’s methenamine silver-staining, end-point PCR, RT-PCR, and beta-D-glucan assay
Results:
All the microscopically positive bronchoalveolar-lavage samples (50%) also resulted positive by end-point and real time PCR and all, but two, resulted positive also by beta-D-glucan quantification. End-point PCR and RT-PCR detected 10 (55%) and 11 (61%) out of the 18 samples, respectively, thus showing an enhanced sensitivity in comparison to microscopy. All RT-PCR with a Ct< 27 were confirmed microscopically, whereas samples with a Ct≥ 27 were not.
Conclusions:
Our work highlights the need of reshaping and redefining the role of molecular diagnostics in a peculiar clinical setting, like P. jirovecii infection, which is a rare but also severe and rapidly progressive clinical condition affecting immunocompromised hosts that would largely benefit from a faster diagnosis. Strictly selected patients, according to the inclusion criteria, resulting negative by molecular methods could be ruled out for P. jirovecii pneumonia.

Franconi I, Leonildi A, Erra G, et al. Comparison of different microbiological procedures for the diagnosis of Pneumocystis jirovecii pneumonia on bronchoalveolar-lavage fluid. BMC Microbiol. 2022;22(1):143. Published 2022 May 21. doi:10.1186/s12866-022-02559-1
Post time: Jun-14-2022
